
 |

|
|
||||||||||||||||||||||
THE SYSTEM OF NON-APPARATUS MEMBRANE PLASMAPHERESIS HANDLED WITH A SYRINGE UBM-01-"PF SPb"
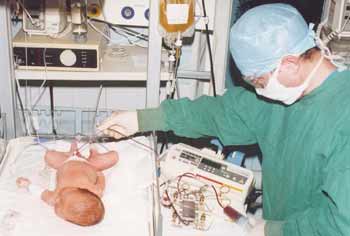 One of the main causes of the severe neonatal illness and infant mortality is a syndrome of endogenous intoxication, occuring in acute pneumonia, hemolytic disease, hyperbilirubinemia, septic and other complications, when even new antibiotics are helpless. Implementation of detoxification procedures by generally accepted methods of efferent therapy (hemosorbtion and plasmapheresis, - gravity, apparatus or packed) to newborns, especially to premature babies, faces several technical and methodical problems.
One of the main causes of the severe neonatal illness and infant mortality is a syndrome of endogenous intoxication, occuring in acute pneumonia, hemolytic disease, hyperbilirubinemia, septic and other complications, when even new antibiotics are helpless. Implementation of detoxification procedures by generally accepted methods of efferent therapy (hemosorbtion and plasmapheresis, - gravity, apparatus or packed) to newborns, especially to premature babies, faces several technical and methodical problems.
The main difficulties occur because of: •low birth weight and, consequently, small volume of circulating blood; •instability of hemodynamics that is very sensitive to the deficiency of circulating blood volume; •limitations in using catheters with a diameter of more than 0.6 - 0.8 mm; •long-term location of catheters in the umbilical vessels, that might results in thrombosis of the portal system and various septic complications. Most of known methods of plasmapheresis have a number of limitations: too large priming volume comparable, for example, with a total blood volume of neonate and have the perfusion rate far exceeding capability of small vessels and catheters. Method of blood sampling by a syringe (up to 20 ml) followed by centrifugation, when plasma is discharged and packed red blood cells reinfused, are very tedious and time consuming (up to 4 - 6 hours). Significant difficulties posesses the plasmapheresis on infants and on children weighing less than 15 - 20 kg. Maximum allowable single blood sampling in these patients should not exceed 100 - 150 ml, that make impossible using the most common centrifuge bag/batch methods. JSC "Plasmofilter" (St. Petersburg) has developed membrane plasma filters PFM-800 (PFM-500), mass production of which was authorized in 1992 by the Commission on New Medical Technology of Russian Ministry of Health. The membrane plasma filters meet the requirements for the plasmapheresis procedures in newborns, premature babies and young children because of minimum priming volume (less than 15 ml) and can be used without any devices with just one (or two) syringes for one-needle scheme technique. The system includes the membrane plasma filter PFM-500 (or PFM-800) and transfusion line working with a syringe.  The system of non-apparatus membrane plasmapheresis UMB-01-"PF SPb" handled with a syringe. The method and the device are unique and have no analogues in the practice. Plasmapheresis procedure is performed using one-needle scheme with a disposable syringe. At the first phase (exfusion) blood is withdrawed into the syringe. At the second phase (reinfusion) blood is passed through the plasma filter, where a portion of plasma is separated and the packed red blood cells are sent back into the vein. Small priming volume (as little as 10 ml) allows to perform plasmapheresis even on premature babies weighing 700 — 800 g.
The method and the device are protected by RUS patent No. 2113863: "Method of membrane plasmapheresis using a one-needle scheme and device for its implementation".
Ease of use the plasma filters with various apparatus, rollers or even in stand alone mode (using a gravity force only) make the procedures of membrane plasmapheresis available practically for any health facility. |
|||||||||||||||||||||||
|
|
|
||||||||
Leninskiy pr., bld.140, Saint Petersburg, 198216, Russia, tel. +7 (812) 376-9079, +7 (812) 376-9070; +7 (921) 9168147 mob.; fax: +7 (812) 376-9079 |
E-mail: plasma01@yandex.ru www-page: http://www.plasmafilter.sp.ru |
||||||||
| ©ZAO "PLASMOFILTER", 2023 Design and maintenance: Eugene Vovenko |
Last updated: | ||||||||